The Federal Drug & Food Administration is at it again. In recent moves to help change the tide of obesity in America, the FDA has officially banned trans fats from foods. They’ve also proposed an idea that puts the Percent Daily Value (%DV) of sugar on nutrition labels. So what’s next for them? A temporary, non-surgical balloon, placed in the stomach to help combat obesity.
The FDA has officially approved this balloon device to be used instead of invasive surgery. The ReShape Dual Balloon is exactly what it sounds like. Two, saline filled, attached balloons are inserted through the mouth, making it a great choice for those who hate or don’t want surgery. These balloons take up room in the stomach, giving the person a feeling of fullness, as well as less room to eat. That’s also the idea behind two balloons instead of one; more occupied space. The maximum period of time to have this device is six months, after which it is removed by a medical professional.
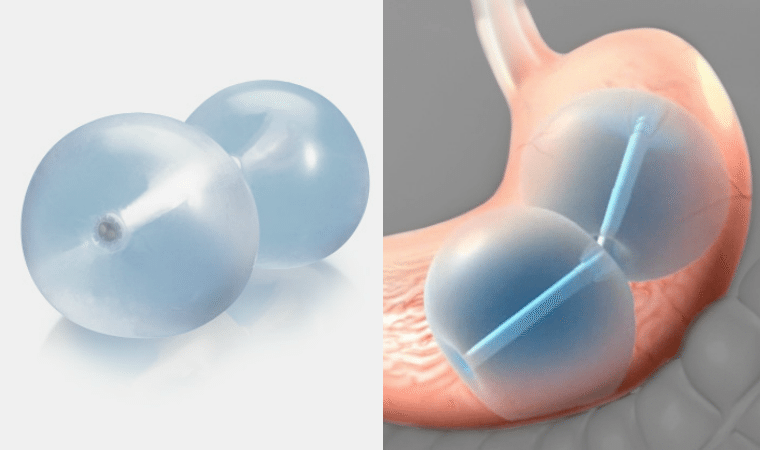
This method of weight loss is intended for people who are overweight and have a Body Mass Index (BMI) between 30 and 40 kg/m2 . It is only available to patients with one or more conditions associated with obesity, such as high cholesterol and diabetes. It should be noted that this device is also only intended for obese adults who have failed weight loss with diet and exercise alone. Unfortunately, for people with gastrointestinal issues, the occupation of the balloons could cause intestinal block and irritation, therefore it is not allowed for anyone with GI problems.
Clinical trials studied 326 obese adults, ranging from ages 22-60. 187 of them were randomly selected to have the balloon procedure done, while the others were used as a control group. After six months, the group that had the ReShape Dual balloons lost on average 14.3 pounds, roughly 6.8 percent of their total body weight. The control group only lost an averaged 7.2 pounds, which is 3.3 percent of their body weight. In addition, the participants who had the procedure done were followed up with six months after the study concluded. Said participants were able to keep off an average of 9.9 pounds of the 14.3 they lost.
But it’s not all happiness and weight loss one these balloons are implanted. The FDA says that after the procedure takes place, patients may experience vomiting, nausea, abdominal pain, gastric ulcers, and feelings of indigestion. Now, that doesn’t sound like a fun time.
The FDA has already approved other obesity combating procedures such as Allergan LAP-Band, the Ethicon Endo-Surgery Realize Adjustable Gastric Band, and the Maestro Rechargeable System. But what makes ReShape different? It’s its lack of invasive surgery and that little six month time stamp. This makes the device a helping hand in a weight loss journey instead of a new, permanent fixture in their body. More still has to be done to put this product on the market, but the future looks promising for users of this weight loss method.
For more on Work & World, check out our articles here.
What do you think of the FDA’s move to approve this implant?








![Daily Bite [Make]: Philly Cheesesteak Stuffed Bell Peppers](https://dashofwellness.com/wp-content/uploads/2013/01/Philly-Cheesesteak-Stuffed-Pepper-Daily-Bite-1-100x70.png)
